Page Contents
ToggleProcess to Import Cosmetics from USA to India – CDSCO Registration Guide
India has become one of the fastest-growing beauty and personal care markets in the world. As a result, many global companies want to import cosmetics from USA to India to reach Indian consumers. American skincare, makeup, and wellness brands already enjoy strong demand in India because consumers often trust international product standards.
However, cosmetic products cannot be directly shipped and sold in India without regulatory approval. Before companies can import cosmetics to India from USA, they must comply with Indian cosmetic regulations.
Under the Cosmetics Rules 2020, all imported cosmetic products require CDSCO cosmetic import registration before entering the Indian market. The Central Drugs Standard Control Organization (CDSCO) regulates cosmetic imports and ensures product safety, labeling compliance, and ingredient transparency.
Therefore, both US cosmetic manufacturers and Indian importers must complete CDSCO registration before selling cosmetic products in India.
This guide explains the complete process for importing cosmetics from USA to India, including regulatory requirements, documentation, government fees, and compliance guidelines.
Quick Overview – Import Cosmetics from USA to India
Authority: CDSCO (Central Drugs Standard Control Organization)
Application Form: COS-1
Registration Certificate: COS-2
Government Fee: $1000 per category + $500 per manufacturing site + $50 per variant
Approval Timeline: 60-90 days
Experts like Regacats Solutions can help you get Cosmetic import registration (Cos-2) Certificate in 30-35 days.
This quick overview helps importers and foreign manufacturers understand the basic requirements for CDSCO cosmetic registration USA products.
Is CDSCO Registration Required to Import Cosmetics from USA to India?
Yes. Any company planning to import cosmetics from USA to India must obtain a COS-2 Cosmetic Import Registration Certificate from CDSCO.
CDSCO regulates imported cosmetics under the Cosmetics Rules 2020 to ensure safety and product quality.
The registration process generally works as follows:
The applicant submits a COS-1 application through the CDSCO SUGAM portal
CDSCO reviews the submitted documentation
CDSCO may raise regulatory queries if clarification is needed
After approval, CDSCO issues the COS-2 Registration Certificate
Without COS-2 approval, cosmetic products cannot legally enter India through customs.
Therefore, CDSCO cosmetic registration for US brands is mandatory before distribution or sale in India.
Who Can Apply for Cosmetic Import Registration?
Before companies can import US cosmetics to India, they must identify an eligible applicant.
According to CDSCO regulations, the following entities can apply for cosmetic import registration.
Authorized Indian Agent
Foreign manufacturers often appoint an Authorized Indian Agent for cosmetics export to manage regulatory communication with CDSCO.
Indian Importer
An Indian distributor or importing company may apply for cosmetic registration on behalf of the manufacturer.
Indian Subsidiary of Foreign Manufacturer
If the US cosmetic brand operates an Indian subsidiary, the company may directly submit the application.
However, CDSCO requires a Power of Attorney from the manufacturer authorizing the applicant to represent the brand during the registration process.
CDSCO Registration Process for Importing Cosmetics from USA to India
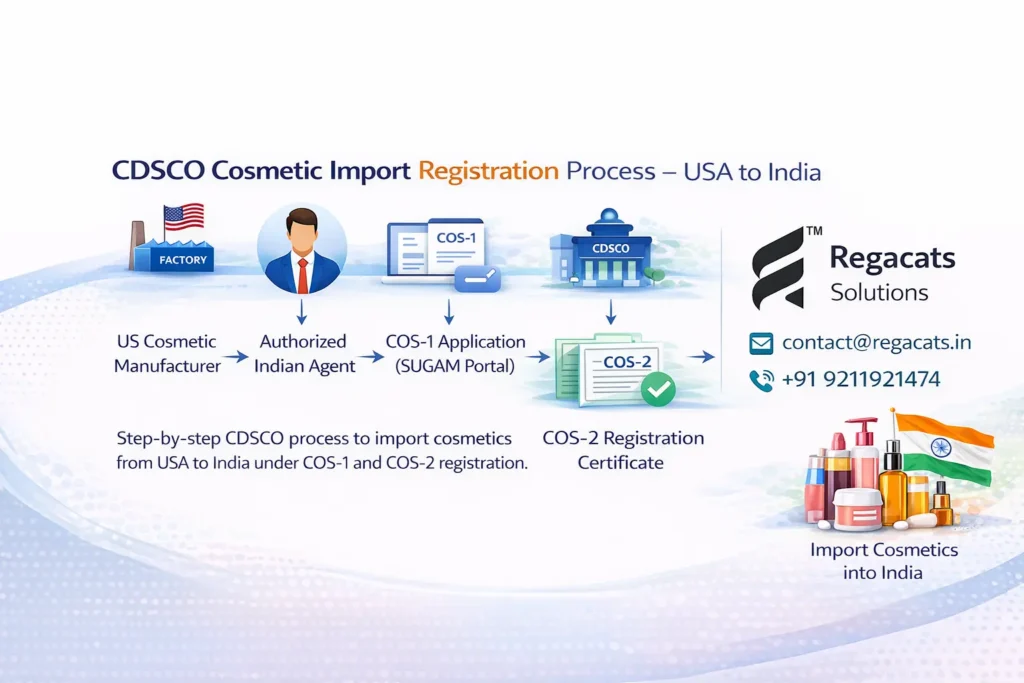
Companies planning to import cosmetics from USA to India must follow a structured CDSCO registration process.
Step 1 – Appoint Authorized Indian Agent
Foreign cosmetic brands usually appoint an Indian regulatory representative to manage the application process.
Step 2 – Create SUGAM Portal Account
Applicants must register on the CDSCO SUGAM online portal, which manages cosmetic import applications.
Step 3 – Submit COS-1 Application
Applicants submit the COS-1 cosmetic registration application along with all required documents.
Step 4 – CDSCO Review
CDSCO evaluates the submitted documents and may request additional clarification.
Step 5 – Receive COS-2 Registration Certificate
After successful evaluation, CDSCO grants the COS-2 cosmetic import registration certificate, allowing legal import and sale in India.
Checklist for Importing Cosmetics from USA to India
To simplify the process, companies can follow this checklist before submitting their CDSCO application.
Prepare Power of Attorney from manufacturer
Obtain Free Sale Certificate
Create SUGAM portal account
Submit COS-1 application
Pay government registration fee
Respond to CDSCO regulatory queries
Receive COS-2 cosmetic import registration certificate
This checklist helps both foreign manufacturers and Indian importers prepare for CDSCO registration.
Documents Required for Importing Cosmetics from USA to India

Applicants must prepare several documents before applying for CDSCO cosmetic registration USA products.
Commonly required documents include:
Power of Attorney from manufacturer
Free Sale Certificate issued by US authority
Ingredient list and formulation details
Product specifications
Manufacturing license
Cosmetic label artwork
Product composition sheet
Product testing data (if required)
Accurate documentation helps avoid CDSCO regulatory queries and approval delays.
Before submitting the COS-1 application, applicants should carefully review the complete documentation framework to prevent missing information or inconsistencies. For a comprehensive overview of all required documents, formatting guidelines, and regulatory checkpoints, refer to our guide on Documents Required for Cosmetic Import License – CDSCO Checklist. This guide explains each requirement in detail and helps ensure that your application is properly prepared and submitted without deficiencies.
Government Fee for Cosmetic Import Registration in India
Companies must pay government fees when applying to import cosmetics from USA to India.
Standard CDSCO fees include:
USD 1000 per category
- USD 500 per manufacturing site
USD 50 per product variant
For example, cosmetic products with multiple shades or variants require separate registration fees.
Therefore, companies should plan their CDSCO cosmetic registration process carefully to optimize regulatory costs.
efore submitting the COS-1 application, applicants should clearly understand the applicable government charges for cosmetic import registration. The CDSCO requires specific fees for product category, manufacturing site and product variants under the Cosmetics Rules 2020. For a detailed explanation of the complete fee structure, payment process, and cost calculation for multiple product variants, refer to our guide on Cosmetic Import Registration Fees in India (COS-1 & COS-2). This guide provides a clear breakdown of government fees and helps importers plan the registration cost more effectively before starting the CDSCO application process.
CDSCO Cosmetic Labeling Requirements in India
Cosmetic products imported into India must follow labeling rules under the Cosmetics Rules 2020.
Labels must include:
Product name
Manufacturer name and address
Importer name and address
Batch or lot number
Manufacturing or expiry date
Ingredient list
Proper labeling ensures transparency and consumer safety.
Therefore, companies planning to import cosmetics to India from USA should verify label compliance before shipping products.
Timeline for Cosmetic Import License in India
The approval timeline for cosmetic import license India usually depends on documentation quality and CDSCO review.
In most cases, approval takes 60 to 90 days.
However, delays may occur if:
CDSCO raises regulatory queries
documents require corrections
labeling changes are necessary
Proper documentation helps reduce approval delays.
Experienced CDSCO consultants (Regacats Solutions) help foreign manufacturers, global brands, importers get cosmetic import registration (Cos-2) certificate in 30-35 days.
Common Challenges When Importing Cosmetics from USA to India
Many companies encounter regulatory and documentation challenges when they try to import cosmetics from USA to India. These issues usually arise during the CDSCO review process and can delay approval if companies do not address them properly.
Some of the most common challenges include the following.
Labeling Non-Compliance for Importing Cosmetics from USA to India
Labels created for the US market often follow FDA labeling practices. However, these labels may not fully comply with Indian cosmetic labeling requirements under the Cosmetics Rules 2020. Importers must ensure that the label includes the importer’s details, batch number, manufacturing or expiry date, and ingredient information according to Indian regulations.
Missing or Incomplete Free Sale Certificate
CDSCO requires a valid Free Sale Certificate (FSC) issued by the competent authority in the country of origin. If the FSC is missing, expired, or incorrectly issued, CDSCO may raise queries that delay the approval process.
Ingredient Compliance Issues
Some cosmetic ingredients permitted in other countries may require additional regulatory verification in India. CDSCO may request further clarification if ingredient declarations are incomplete or inconsistent with regulatory standards.
CDSCO Queries During COS-1 Application Review
When companies import cosmetics from USA to India, CDSCO may raise queries during the COS-1 application review. These queries commonly relate to ingredient inconsistencies, incomplete or improperly apostilled Power of Attorney, non-compliant labeling artwork, or documentation gaps.
If applicants do not respond correctly, these issues can significantly extend the cosmetic registration approval timeline.
To understand these risks in more detail, you can review our guide on CDSCO cosmetic import registration deficiencies, which explains the most frequent regulatory queries and how companies can avoid delays during the approval process.
Why Choose Regacats Solutions for Importing Cosmetics from USA to India?
Entering the Indian cosmetics market requires strong regulatory planning and compliance with CDSCO rules.
Regacats Solutions provides professional cosmetic import registration consulting services in India to help companies import cosmetics from USA to India smoothly and efficiently.
Our regulatory experts assist with:
CDSCO cosmetic import registration (COS-1 & COS-2)
regulatory consulting for foreign cosmetic brands
documentation preparation and review
labeling compliance under Cosmetics Rules 2020
Authorized Indian Agent support
CDSCO query handling
By working with Regacats Solutions, foreign manufacturers and importers can complete CDSCO cosmetic registration efficiently while avoiding regulatory delays.
For a detailed understanding of documentation requirements, eligibility conditions, government fees, and approval timelines, you can explore our Cosmetic Import Registration Guide. The guide outlines the complete CDSCO cosmetic import registration process and explains how regulatory authorities assess foreign-manufactured cosmetic products before approval.
FAQs – Importing Cosmetics from USA to India
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import registration, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import registration, and FSSAI regulatory consulting in India.
Conclusion
India offers strong growth opportunities for international beauty brands. However, companies must complete CDSCO cosmetic registration before they can import cosmetics from USA to India.
By understanding CDSCO regulations, documentation requirements, government fees, and labeling rules, both US manufacturers and Indian importers can successfully launch cosmetic products in the Indian market.
With proper regulatory planning and expert support from Regacats Solutions, foreign cosmetic brands can enter India while maintaining full compliance with Cosmetics Rules 2020.
Ready to launch in India?
Get COS-2 registration services and start import.
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
















