Page Contents
ToggleWhat Are Cosmetic Import Registration Deficiencies?
Cosmetic import registration deficiencies refer to documentation errors, labeling non-compliance, or regulatory inconsistencies identified by CDSCO during review of a COS-2 application. These deficiencies often delay approval timelines and require structured clarification.
Therefore, understanding common deficiencies before submission helps importers avoid approval delays and rejection risks.
For a broader understanding of the overall approval framework, eligibility requirements, and step-by-step process, refer to our detailed guide on CDSCO cosmetic license for importer (COS-1 & COS-2). That guide explains the complete regulatory pathway before and after submission of Form COS-1.
Understanding common CDSCO Cosmetic Import Registration Deficiencies helps businesses secure faster approvals and avoid unnecessary compliance risks.
Common Cosmetic Import Registration Deficiencies in COS-2 Applications

1. Incorrect or Incomplete Ingredient Declaration
One of the most frequent deficiencies involves ingredient mismatch. CDSCO cross-verifies the ingredient list with:
Product label artwork
Product specification sheet
Free Sale Certificate
If names differ, concentrations appear inconsistent, or restricted substances raise concern, CDSCO issues a query immediately.
Therefore, ensure complete alignment across all documents before submission.
2. Improper Power of Attorney (PoA)
Foreign manufacturers must issue a properly notarized and apostilled Power of Attorney in favor of the Authorized Indian Agent.
Common mistakes include:
Missing apostille
Incorrect company details
Expired authorization
Improper signatory designation
As a result, the application remains pending until corrected documents are submitted.
3. Label Non-Compliance with Indian Regulations
Imported cosmetic labels must comply with the Cosmetics Rules, 2020. CDSCO often raises objections regarding:
Missing importer address
Absent COS-2 registration number
Incorrect ingredient order
Missing manufacturing/expiry details
Consequently, artwork revision becomes necessary before approval.
4. Incomplete Manufacturing Details
CDSCO verifies:
Manufacturing site address
GMP certificate
Product category alignment
If manufacturing details do not match across documents, regulatory officers request clarification.
5. Incorrect Product Categorization
Sometimes applicants group products under incorrect cosmetic categories. However, CDSCO evaluates classification carefully. Misclassification leads to re-evaluation and delay.
6. Missing or Improperly Issued Free Sale Certificate is one of the main Cosmetic import registration deficiencies
The Free Sale Certificate must:
Be issued by a competent authority
Clearly list the product
Remain valid at submission time
If validity lapses, CDSCO requires fresh documentation.
How to Avoid Cosmetic Import Registration Deficiencies
Avoiding cosmetic import registration deficiencies requires proactive compliance planning rather than reactive correction. Many importers submit COS-1 applications without conducting a structured document review. As a result, CDSCO identifies inconsistencies that delay approval.
To prevent deficiencies, follow these critical steps:
1. Conduct a Pre-Submission Document Audit
Before filing, verify that ingredient declarations match across the label artwork, product specification sheet, and Free Sale Certificate. Even minor naming variations can trigger queries.
2. Validate Ingredient Compliance
Check that all ingredients comply with the Cosmetics Rules, 2020. Restricted or borderline ingredients often lead to scrutiny. Therefore, review concentration levels and regulatory status carefully.
3. Ensure Properly Executed Power of Attorney
Foreign manufacturers must issue an apostilled and clearly authorized Power of Attorney. Missing apostille, incorrect company details, or improper signatory designation commonly result in deficiency letters.
4. Review Label Artwork for Indian Compliance
Confirm that the label includes:
Importer name and address
Manufacturing details
Batch number
Use-before date
Space for COS-2 registration number
Small labeling gaps frequently cause avoidable delays.
5. Cross-Check Product Categorization
Incorrect product classification under cosmetic categories often forces CDSCO to seek clarification. Align your product type accurately before submission.
6. Perform Regulatory Alignment Review
A structured compliance review significantly reduces query risk. Many companies therefore engage a Cosmetic import registration consultant in India to validate documentation before filing.
When businesses take preventive steps instead of reacting to deficiency letters, approval timelines improve substantially.
Why Cosmetic Import Registration Deficiencies Cause Approval Delays
Cosmetic import registration deficiencies directly impact approval timelines because CDSCO processes applications sequentially. When officers detect inconsistencies, they issue a formal query and pause evaluation until the applicant responds.
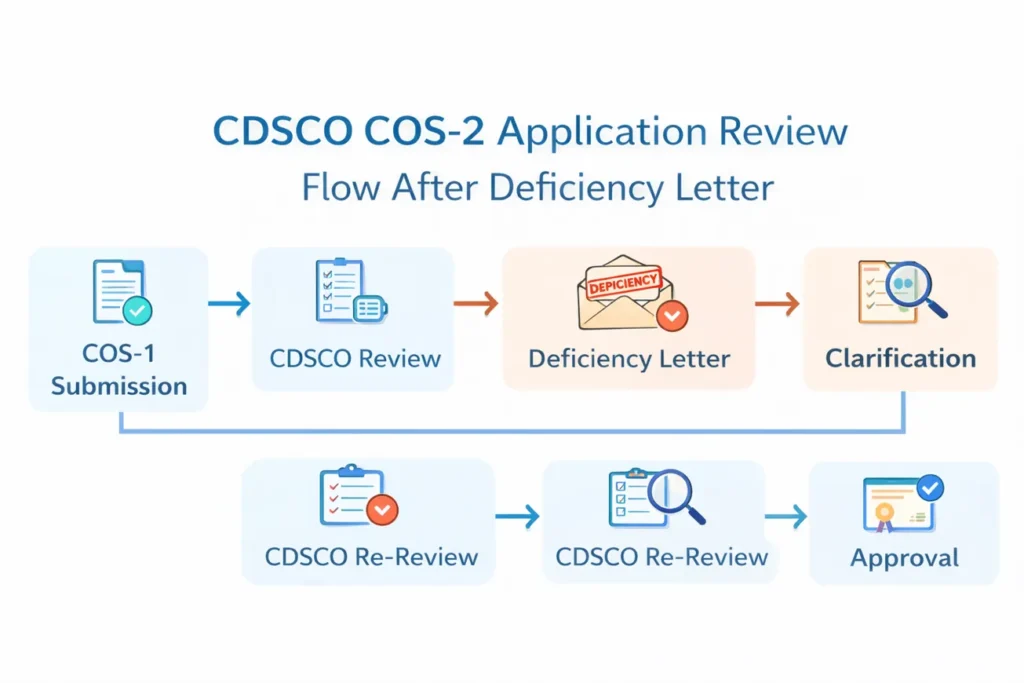
Several factors contribute to delays:
1. Regulatory Queue Reset
Once CDSCO issues a deficiency letter, the application effectively re-enters the review cycle after clarification submission. Consequently, processing time extends by several weeks.
2. Document Re-Verification
Officers re-examine the entire file after receiving revised documents. Even if the correction appears minor, they reassess ingredient alignment, labeling compliance, and manufacturing details again.
3. Increased Scrutiny After Multiple Queries
If an applicant submits incomplete responses repeatedly, regulatory confidence declines. As a result, scrutiny increases, and approval slows further.
4. Port-Level Implications
In some cases, pending deficiencies delay customs clearance because registration approval remains incomplete. This can disrupt market launch plans and distributor commitments.
5. Commercial Impact
Extended approval timelines affect inventory planning, marketing schedules, and brand entry strategy in India. Therefore, preventing cosmetic import registration deficiencies becomes a strategic business priority rather than a procedural formality.
For a complete understanding of the overall approval pathway, review our detailed guide on CDSCO cosmetic license for importer (COS-1 & COS-2), which explains the full regulatory framework governing cosmetic imports into India.
How to Avoid Cosmetic Import Registration Deficiencies
Regulatory queries typically extend approval timelines by 30–60 days. Moreover, repeated deficiencies affect importer credibility.
Therefore, businesses planning to import cosmetics into India should conduct a structured pre-submission compliance review before filing COS-1.
Working with an experienced Cosmetic import registration consultant in India helps identify documentation gaps, labeling inconsistencies, and regulatory risks before CDSCO raises a deficiency letter. As a result, approval timelines improve and unnecessary delays are avoided.
How to Reduce CDSCO Query Risk
To minimize deficiencies:
Conduct internal document cross-verification
Review labeling against Indian compliance rules
Validate PoA and apostille
Confirm ingredient permissibility
Double-check manufacturing documentation
However, even experienced importers face unexpected queries. That is why many businesses engage specialized consultants for end-to-end regulatory handling.
Facing Delay due to Cosmetic Import Registration Deficiences?
If your cosmetic import application has received a deficiency letter, expert review can significantly reduce response time and approval risk. Our regulatory team assists importers and global cosmetic brands with document correction, compliance alignment, and query resolution.
Talk to a CDSCO expert today and ensure smooth approval without repeated delays.
FAQ-Cosmetic Import Registration Deficiencies Cos-2 Application
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import registration, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import registration, and FSSAI regulatory consulting in India.
Conclusion
To ensure safe and easy market entry, consult a qualified regulatory expert. A trusted CDSCO consultant in India like Regacats not only eliminates that risk — but accelerates your growth in one of the world’s fastest-growing beauty markets.
Ready to launch in India?
Get COS-2 registration services and start import.
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
















