Page Contents
ToggleComplete Guide to CDSCO Medical Device Import License in India
If you want to import medical devices into India, you must obtain a medical device import license under CDSCO regulations. This guide explains the complete process, documentation, timeline, and MD15 approval steps.
In this guide, you will learn:
- How to get CDSCO Medical Device import license
Complete medical device import process in India
CDSCO Medical Device Import License (MD 14, MD 15) process step-by-step
Required documents for CDSCO Medical Device Import License (MD 14, MD 15)
Timeline and compliance requirements for CDSCO Medical Device Import License (MD 14, MD 15)
Role of an Indian Authorized Agent in CDSCO Medical Import License (MD 14, MD 15)
Whether you are an Indian importer or a foreign manufacturer, this article explains everything in clear and practical terms.
What is CDSCO Medical Device Import License?
The Central Drugs Standard Control Organization (CDSCO) regulates medical devices in India under the Medical Device Rules, 2017.
If a company wants to import medical devices into India, it must obtain:
MD14 – Application for Medical Device Import License
MD15 – Medical Device Import License issued by CDSCO
Without MD15 approval, customs authorities will not clear the products.
Who Needs a CDSCO Medical Device Import License (MD 15)?
You need a CDSCO import license if:
You are importing medical devices into India
You are a foreign manufacturer selling devices in India
You appoint an Indian Authorized Agent
Your device falls under Class A, B, C, or D category
Additionally, even diagnostic kits, surgical instruments, and electronic medical devices require registration if they fall under notified categories.
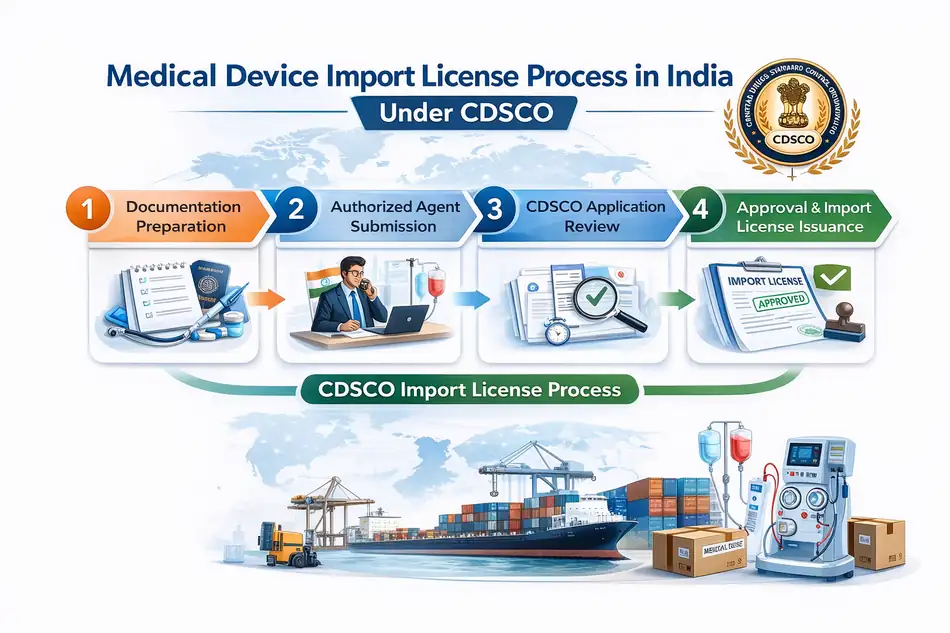
How to Get CDSCO Medical Device Import License (MD 15) in India (Step-by-Step)

Below is the complete medical device import license process in India.
Step 1: Determine Device Classification
First, identify whether your device falls under:
Class A (Low risk)
Class B (Low moderate risk)
Class C (Moderate high risk)
Class D (High risk)
The classification determines review complexity and approval timeline.
Read: Medical Device Classification India: CDSCO Class A B C D Guide
Step 2: Appoint an Indian Authorized Agent (For Foreign Manufacturers)
Foreign manufacturers must appoint an Indian Authorized Agent who:
Holds a valid wholesale drug license
Acts as the official liaison with CDSCO
Submits MD14 application
Responds to regulatory queries
Without an Authorized Agent, CDSCO will not process the application.
If you are a foreign manufacturer looking to appoint an Indian Authorized Agent for CDSCO import licensing, you can explore our CDSCO Medical Device Import License Consultant in India.
Step 3: Prepare Documentation for MD14 Application
You must prepare the following documents:
Covering letter
Power of Attorney
Free Sale Certificate
ISO 13485 certificate
CE certificate (if applicable)
Device Master File
Plant Master File
Label and IFU copies
Undertaking and declarations
Proper documentation reduces query risks significantly.
Step 4: Submit MD14 Application via CDSCO SUGAM Portal
Next, the Indian Authorized Agent submits Form MD14 online through the SUGAM portal.
At this stage:
Government fees must be paid
All technical documents must be uploaded
Accuracy is critical
Even small mistakes can delay approval.
Step 5: CDSCO Review & Query Handling
After submission:
CDSCO reviews technical documentation
Authorities may raise queries
Clarifications must be submitted within the deadline
Proactive query management improves approval speed.
Step 6: Grant of MD15 Import License
If CDSCO approves the application:
MD15 import license is issued
License remains valid perpetually (subject to retention fee)
Importer can start commercial imports
This completes the CDSCO MD15 process.
CDSCO MD15 Process Timeline
The timeline depends on device class:
Class A/B: 3–6 months
Class C/D: 6–9 months
However, incomplete documentation can extend timelines.
Therefore, professional regulatory preparation significantly reduces delays.
Documents Required for CDSCO MD14, MD 15 Import License

Here is a simplified checklist:
Power of Attorney from Manufacturer
ISO 13485 Certificate
Free Sale Certificate
CE Design Certificate (for higher classes)
Device Master File
Plant Master File
Labeling as per MDR 2017
Wholesale License of Authorized Agent
You should prepare documents exactly as per CDSCO format to avoid rejection.
Government Fees for CDSCO MD 14, MD 15 Import License
Government fees depend on:
Device class
Number of Devices
Manufacturing site
Additionally, official CDSCO fee applies per manufacturing site and per device.
Common Reasons for Delay in CDSCO Medical Device Import License (MD 15)
Many applications face delays due to:
Incorrect device classification
Incomplete Device Master File
Improper labeling compliance
Weak clinical data
Inexperienced query response
Therefore, structured regulatory planning is essential.
To avoid regulatory delays, many importers work with experienced CDSCO regulatory consultants who manage documentation and query handling end-to-end.
Role of Indian Authorized Agent in CDSCO Medical Device Import License (MD 15) Process
The Indian Authorized Agent:
Submits application
Maintains regulatory records
Handles post-approval compliance
Manages retention fees
Communicates with CDSCO
Moreover, the agent carries legal responsibility for compliance in India.
Choosing a qualified regulatory partner ensures long-term compliance security.
Can Foreign Manufacturers Apply Directly for CDSCO Import License for Medical Device (MD 15)?
No. Foreign manufacturers cannot directly apply.
They must appoint:
An Indian Authorized Agent
A licensed wholesale distributor
This requirement ensures regulatory accountability inside India.
Post-Approval Compliance Requirements For Imported Medical Device
After receiving MD15:
Maintain updated documentation
Report adverse events
Pay retention fees every five years
Update license for major changes
Failure to comply may lead to suspension.
Frequently Asked Questions on Medical Device Import License
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import registration, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import registration, and FSSAI regulatory consulting in India.
Conclusion
Obtaining a CDSCO medical device import license requires structured planning, accurate documentation, and strong regulatory understanding.
While the process appears straightforward, regulatory queries often delay approvals. Therefore, many importers and foreign manufacturers choose professional guidance to ensure smooth approval
Need Help with CDSCO Import License in India?
If you are an importer or foreign manufacturer planning to register medical devices in India, professional regulatory assistance can simplify the entire MD15 process.
Explore our detailed service offering here:
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
















