Page Contents
ToggleMedical Device Import License Requirements in India – CDSCO Complete Guide
Understanding medical device import license requirements is essential for companies planning to enter the Indian healthcare market. The Central Drugs Standard Control Organization (CDSCO) regulates medical devices under the Medical Device Rules, 2017. Therefore, importers and foreign manufacturers must comply with strict regulatory requirements before selling medical devices in India.
Although many businesses focus only on documentation, the approval process depends on classification, compliance, and regulatory accuracy. Consequently, missing even one requirement can delay approval or trigger CDSCO queries.
Before proceeding, you should review the complete medical device import license guide to un derstand the overall regulatory framework and approval process in India.
Medical Device Import License Requirements in India Under CDSCO
The requirements for Medical Device import in India are defined under the Medical Device Rules, 2017. CDSCO ensures that all notified medical devices meet safety, quality, and performance standards.
To import medical devices into India, applicants must:
- Submit Form MD14 through the CDSCO SUGAM portal
- Provide complete technical documentation
- Appoint an Indian Authorized Agent
- Ensure labeling compliance under MDR 2017
- Maintain quality standards such as ISO 13485
Additionally, CDSCO evaluates each application based on classification and intended use. Therefore, understanding regulatory expectations before submission is critical.
Key Medical Device Import License Requirements Based on Device Classification
Device classification directly influences medical device import license requirements. CDSCO categorizes devices into Class A, B, C, and D based on risk levels.
- Class A – Low risk
- Class B – Low to moderate risk
- Class C – Moderate to high risk
- Class D – High risk
Higher-risk devices require more detailed technical documentation and deeper evaluation. Therefore, companies must determine accurate classification before applying.
To understand classification in detail, refer to medical device classification India, which explains CDSCO risk categories and regulatory implications.
Documentation Requirements for Medical Device Import License
Accurate documentation is one of the most critical requirements for Medical Device import. CDSCO verifies documentation to ensure compliance and product safety.
Required documents include:
- Power of Attorney
- ISO 13485 Certificate
- Free Sale Certificate
- Device Master File (DMF)
- Plant Master File (PMF)
- Labeling compliance as per MDR 2017
- Wholesale license of authorized agent
Incomplete documentation often leads to regulatory delays. Therefore, applicants must prepare documents carefully.
For a detailed checklist, review documents for medical device import to ensure full compliance.
MD14 and MD15 Process for Import License Approval
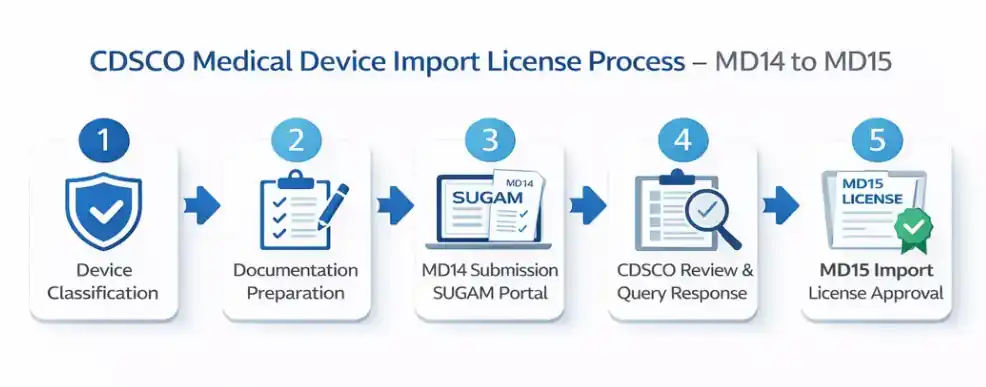
The CDSCO import license process involves multiple stages.
First, applicants submit Form MD14 along with required documentation. Then CDSCO reviews the application and may raise queries. Finally, upon approval, CDSCO issues the MD15 import license.
The MD15 license allows legal import and distribution of medical devices in India.
To understand the differences between application and approval stages, read MD14 vs MD15 CDSCO, which explains the process clearly.
Role of Indian Authorized Agent in Import License Requirements
Foreign manufacturers cannot directly apply for CDSCO import licenses. Therefore, appointing an Indian Authorized Agent is mandatory.
The authorized agent:
- Submits MD14 application
- Communicates with CDSCO
- Ensures compliance
- Handles post-approval responsibilities
Choosing the right representative significantly improves approval success.
To understand this requirement in detail, explore Indian Authorized Agent for medical devices.
Medical Device Import License Requirements for Foreign Manufacturers
Foreign manufacturers must comply with specific requirements for Medical Device import before entering India.
They must:
- Appoint an Indian Authorized Agent
- Provide Power of Attorney
- Submit global certifications (CE/FDA if applicable)
- Ensure manufacturing compliance
Additionally, manufacturers must align all documents with CDSCO expectations. Therefore, proper planning is essential.
Common Mistakes in Medical Device Import License Requirements
Many applicants face delays due to incorrect interpretation of requirements for Medical Device import.
Common mistakes include:
- Incorrect classification
- Incomplete documentation
- Labeling errors
- Weak technical files
- Delayed CDSCO responses
However, companies can avoid these issues with proper preparation and expert guidance.
Timeline and Cost Factors Affecting Import License Approval
Approval timelines depend on classification and documentation quality.
- Class A/B → 3–6 months
- Class C/D → 6–9 months
Additionally, costs vary depending on device complexity and regulatory requirements.
To understand cost breakdown and timelines, refer to medical device import license cost and timeline.
Why Professional Regulatory Support Improves Approval Success
Understanding regulatory requirements can be complex. Therefore, many companies rely on experts for compliance.
Regacats Solutions supports:
- Device classification
- Documentation preparation
- CDSCO submission
- Query handling
- License approval
Businesses can explore medical device import license services in India to streamline the process and reduce approval delays.
FAQs – Medical Device Import License Requirements
Conclusion
Medical device import license requirements define the regulatory pathway for importing medical devices into India. From classification to documentation and approval, every step requires accuracy and compliance.
Companies that follow these requirements properly achieve faster approvals and avoid delays. Therefore, businesses entering the Indian market must focus on regulatory planning and compliance.
With expert guidance and proper documentation, companies can successfully obtain CDSCO import licenses and expand into India.
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import License, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import License, and FSSAI regulatory consulting in India.
















