Page Contents
ToggleCDSCO MD-14 vs MD-15: Complete Guide for Medical Device Import License in India
If you plan to import medical devices into India, you will encounter two important terms: MD 14 vs MD 15 CDSCO . However, many importers and foreign manufacturers feel confused about their roles.
So, what exactly is the difference? More importantly, which one do you need?
In this guide, we will clearly explain CDSCO MD 14 & MD 15, including their purpose, process, and how to avoid costly mistakes. Moreover, this article will help you take the next step toward obtaining your CDSCO import license smoothly.
Learn more about our complete Medical Device Import License India.
What is MD-14 in CDSCO?
MD-14 is the application form submitted to CDSCO for obtaining a medical device import license.
In simple terms, it is the first step in the licensing process. The importer or Indian Authorized Agent files this form through the CDSCO Sugam portal.
Key points:
It is an application, not a license
Submitted by importer or authorized agent
Includes all technical and regulatory documents
What is MD-15 in CDSCO?
MD-15 is the official import license issued by CDSCO after approval of MD-14.
Therefore, once CDSCO reviews and approves your application, it grants MD-15, which allows you to legally import medical devices into India.
Key points:
It is the final license
Issued by CDSCO
Mandatory for importing notified medical devices
MD 14 vs MD 15 CDSCO: Key Differences
Understanding MD 14 vs MD 15 CDSCO is essential for every importer.
| Feature | MD-14 | MD-15 |
|---|---|---|
| Type | Application | License |
| Purpose | Apply for import license | Approval to import |
| Submitted by | Importer/Agent | Issued by CDSCO |
| Stage | Initial | Final |
In short, MD-14 starts the process, while MD-15 completes it.
When Do You Need MD 14 vs MD 15 CDSCO?
You will need both forms when importing notified medical devices into India.
Situations where both are required:
Foreign manufacturers exporting to India
Indian importers sourcing devices internationally
Distributors dealing in regulated devices
Important note:
First, you must file MD-14. After that, CDSCO evaluates your application and issues MD-15.
MD 14 vs MD 15 CDSCO Process (Step-by-Step)
Here is a visual representation of MD 14 vs MD 15 CDSCO process:
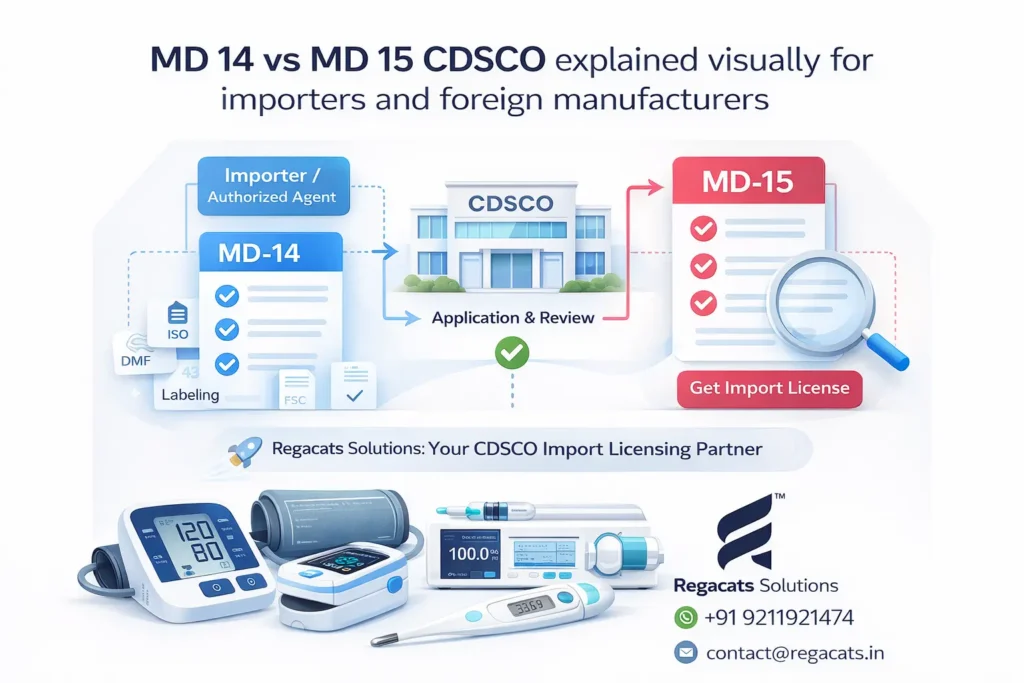
This diagram simplifies the CDSCO import licensing process for better understanding and faster decision-making.
Now let’s understand the complete process of CDSCO MD 15 License.
Step 1: Appoint an Indian Authorized Agent
Foreign manufacturers must appoint a Indian representative to import Medical Device.
Step 2: Prepare documentation
You need:
Free Sale Certificate (FSC)
ISO certification
Device master file
Power of Attorney
Also read our detailed guide on MD-14 documents checklist.
Step 3: Submit MD-14
Upload all documents on the Sugam portal.
Step 4: Review by CDSCO
CDSCO evaluates technical and regulatory compliance.
Step 5: Grant of MD-15
After approval, CDSCO issues the import license.
Common Mistakes in MD 14 CDSCO Applications
Many applications face delays or rejection due to avoidable errors.
Avoid these mistakes:
Incorrect device classification
Incomplete documentation
Errors in Power of Attorney
Missing labeling compliance
Moreover, even small mistakes can delay approval by weeks.
Check our CDSCO Medical Device classification guide to understand Device Classification.
MD 14 vs MD 15 CDSCO: Timeline & Approval Duration
The timeline depends on device class and documentation quality.
Typical timeline:
Class A & B: 30–45 days
Class C & D: 60–90 days
However, incomplete applications can significantly increase processing time.
CDSCO MD 14 & MD 15 for Foreign Manufacturers
Foreign manufacturers must follow additional requirements.
Key requirements:
Appoint Indian Authorized Agent
Submit apostilled documents
Ensure compliance with Indian labeling rules
Why this matters:
Without proper documentation, CDSCO may reject the application.
Frequently Asked Questions
Conclusion
To summarize, understanding CDSCO MD 14 & MD 15 is crucial for anyone planning to import medical devices into India. While MD-14 initiates the process, MD-15 grants legal permission to import.
Therefore, proper documentation and expert guidance can help you avoid delays and ensure faster approval.
Get Expert Help for CDSCO MD-14 & MD-15
If you want to avoid rejection and speed up your approval:
Regacats provides end-to-end CDSCO import license support
We help with:
Documentation preparation
Authorized Agent services
Complete MD-14 & MD-15 application
Contact us today for a FREE consultation
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import License, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import License, and FSSAI regulatory consulting in India.
















