Page Contents
ToggleMedical Device Classification in India – CDSCO Class A, B, C, and D Explained
Understanding medical device classification in India is essential for companies planning to import medical devices into the Indian healthcare market. The Central Drugs Standard Control Organization (CDSCO) regulates medical devices under the Medical Device Rules, 2017. Therefore, classification determines the regulatory pathway, documentation requirements, and approval timeline for obtaining an import license.
Although many manufacturers initially focus on documentation and licensing steps, classification actually defines the entire regulatory framework. Consequently, incorrect classification may lead to regulatory queries, delays in approval, or even rejection of the import license application.
In this guide, we explain:
The regulatory framework of medical device classification in India
CDSCO risk classes (Class A, B, C, and D)
Examples of devices under each category
How classification affects import license requirements
Common classification mistakes
Foreign manufacturers and Indian importers must clearly determine device classification before submitting Form MD14 through the CDSCO SUGAM portal. Proper classification allows companies to prepare accurate regulatory documentation and achieve smoother approvals.
Before determining device classification, manufacturers should clearly understand the overall CDSCO regulatory framework. You can review our detailed medical device import license guide to understand the complete MD14 and MD15 application process, documentation requirements, and approval steps for importing medical devices into India.
Medical Device Classification in India Under Medical Device Rules 2017
The medical device classification in India framework follows a risk-based system established under the Medical Device Rules, 2017. CDSCO evaluates devices according to their potential risk to patients, healthcare professionals, and users.
The classification system includes four categories:
Class A – Low risk
Class B – Low to moderate risk
Class C – Moderate to high risk
Class D – High risk
Each category carries different regulatory requirements, documentation obligations, and evaluation procedures.
Moreover, CDSCO developed this classification framework to align with global regulatory standards. Consequently, the system resembles classification models used by the US FDA and European regulatory authorities.
However, manufacturers must still analyze device characteristics carefully because classification decisions depend on multiple technical factors.
Risk-Based Approach in Medical Device Classification in India
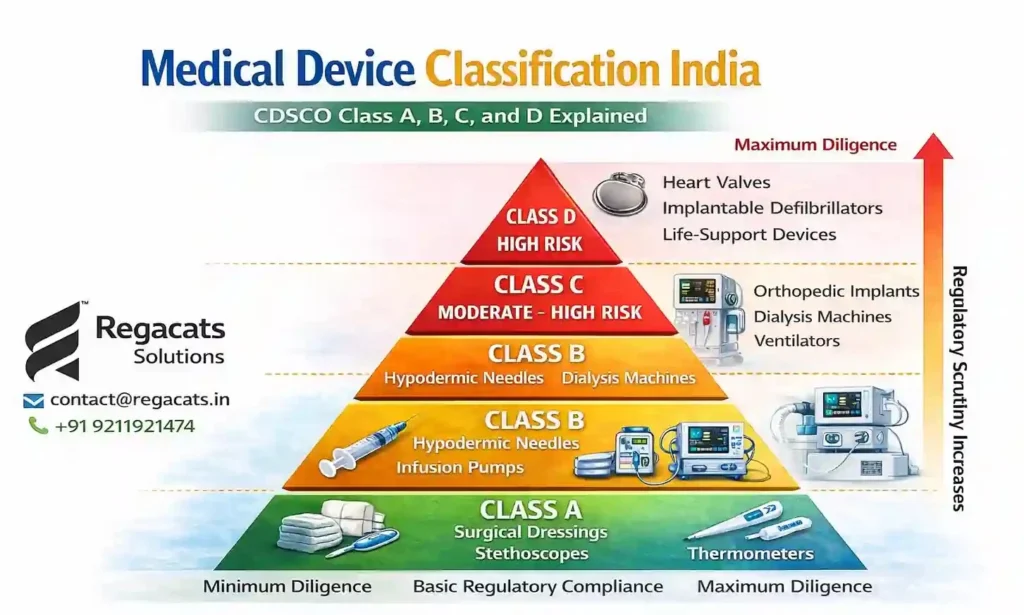
The medical device classification in India system relies on a risk-based regulatory model. CDSCO determines classification by evaluating how a device interacts with the human body and the potential risks associated with its use.
Several factors influence classification:
Duration of device contact with the body
Level of invasiveness
Whether the device sustains life
Intended use of the device
Potential consequences of device malfunction
For instance, simple diagnostic tools generally involve minimal risk and therefore fall under Class A or Class B. However, implantable medical devices present higher risks and fall under Class C or Class D.
As a result, regulatory scrutiny increases with the risk level.
Class A Medical Device Classification
Class A represents the lowest risk level within medical device classification in India.
Devices in this category typically perform simple functions and involve minimal patient risk.
Examples include:
Surgical dressings
Stethoscopes
Thermometers
Hospital beds
Basic diagnostic instruments
Because these devices pose minimal risk, CDSCO applies a relatively straightforward regulatory process. Nevertheless, manufacturers must still comply with essential documentation requirements.
These requirements include:
Device Master File (DMF)
Plant Master File (PMF)
ISO 13485 certificate
Labeling compliance
Therefore, even low-risk devices must meet regulatory standards before receiving approval.
Class B Medical Device Classification
Class B devices fall under the low-to-moderate risk category in the medical device classification India system.
These devices often interact with the human body more directly compared to Class A products.
Examples include:
Hypodermic needles
Infusion pumps
Suction equipment
Surgical gloves
Blood pressure monitoring devices
Because these devices involve greater risk than Class A products, CDSCO performs a more detailed evaluation of documentation.
Applicants must submit technical information demonstrating product safety and manufacturing quality.
Therefore, accurate documentation becomes critical for successful approval.
Class C Medical Device Classification
Class C devices represent moderate-to-high risk medical devices.
Examples include:
Orthopedic implants
Dialysis machines
Ventilators
Bone fixation systems
Infusion systems used in intensive care
Because these devices play an important role in medical treatment, CDSCO performs a detailed technical evaluation before granting approval.
Manufacturers must submit comprehensive documentation such as:
Device Master File
Risk management reports
Clinical evidence where required
Manufacturing process details
Consequently, Class C devices often experience longer review timelines.
Class D Medical Device Classification
Class D devices represent the highest risk category within medical device classification India.
These devices often sustain life or perform critical medical functions.
Examples include:
Implantable cardiac defibrillators
Heart valves
Implantable pacemakers
Life-supporting monitoring systems
Because patient safety risks remain high, CDSCO applies strict regulatory scrutiny.
Manufacturers must provide extensive documentation including:
Clinical evaluation data
Detailed manufacturing validation
Risk management documentation
Post-market surveillance plans
Therefore, Class D applications require thorough regulatory preparation.
Since higher-risk devices require deeper regulatory evaluation, manufacturers must prepare detailed technical documentation before submitting the MD14 application. You can review the complete checklist of documents for medical device import to ensure all regulatory documents comply with CDSCO requirements.
How to Determine Medical Device Classification in India
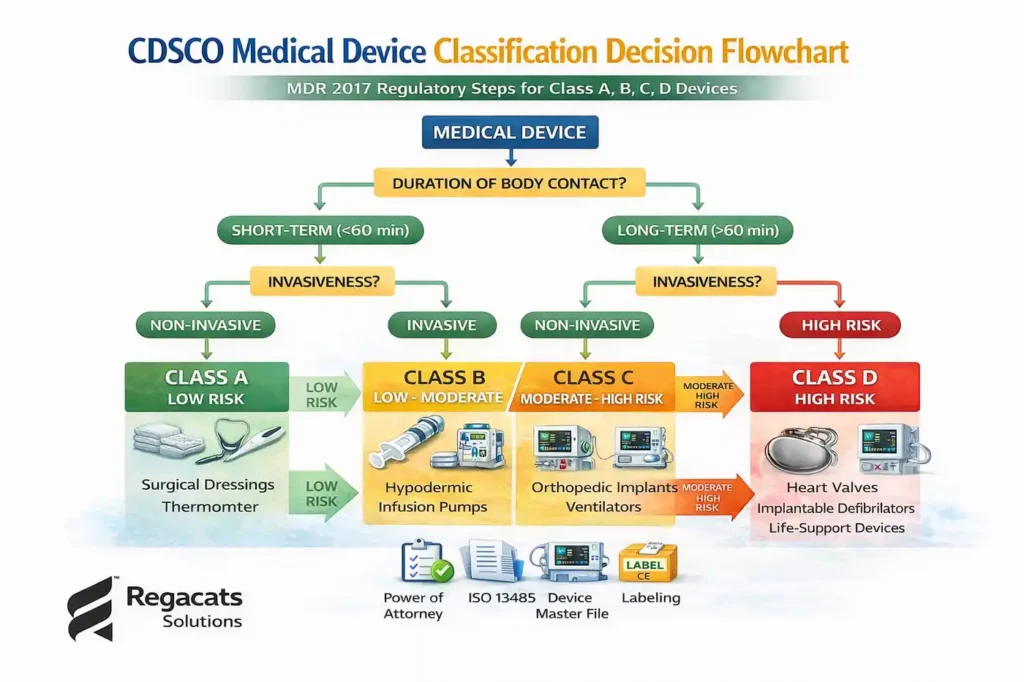
Determining medical device classification in India requires careful analysis of device characteristics.
Applicants must evaluate:
Device functionality
Mode of operation
Contact duration with the body
Level of invasiveness
Risk associated with malfunction
Additionally, manufacturers must review classification rules defined in the Medical Device Rules, 2017.
However, classification decisions sometimes involve complex interpretation. Therefore, regulatory consultants often assist companies in determining accurate classification.
Regulatory Importance of Medical Device Classification in India
The regulatory importance of medical device classification India extends beyond simple categorization.
Classification directly affects:
Import license approval timelines
Technical documentation requirements
CDSCO regulatory review procedures
Compliance obligations for manufacturers
Consequently, incorrect classification may trigger regulatory queries and delay the approval process.
Manufacturers must therefore evaluate classification carefully before submitting the MD14 application.
Role of Regulatory Experts in Device Classification
Foreign manufacturers often encounter challenges when interpreting CDSCO classification guidelines.
Consequently, many companies collaborate with regulatory experts who understand Indian medical device regulations.
Regacats Solutions provides regulatory consulting support for manufacturers planning to import medical devices into India. The team assists companies with device classification review, regulatory documentation preparation, and CDSCO submission procedures.
By working with experienced CDSCO consultants, manufacturers can reduce regulatory risks and improve approval efficiency.
Common Mistakes in Device Classification
Even experienced companies occasionally make classification errors.
Common mistakes include:
Incorrect risk assessment
Misinterpretation of device intended use
Confusion between Class B and Class C devices
Incomplete technical documentation
Ignoring classification rules in MDR 2017
These mistakes frequently lead to CDSCO queries during the review process.
Therefore, companies should conduct a detailed classification assessment before submitting their application.
FAQs – Medical Device Classification
Who determines medical device classification in India?
Manufacturers initially determine device classification based on MDR 2017 rules. However, CDSCO may review and confirm classification during the regulatory evaluation process.
Device classification also influences regulatory review timelines and government fees. For example, higher-risk devices usually require longer technical evaluation. To understand the financial and approval aspects, you can review our guide on medical device import license cost and timeline in India.
Conclusion
Medical device classification India forms the foundation of the CDSCO regulatory framework for medical device imports. The classification system ensures that devices receive appropriate regulatory scrutiny based on their risk level.
Class A and Class B devices follow simplified regulatory procedures, while Class C and Class D devices undergo deeper technical evaluation.
Therefore, manufacturers planning to import medical devices into India must determine device classification carefully before submitting their regulatory application.
Companies that prepare accurate classification documentation significantly improve their chances of faster regulatory approval.
Many companies entering the Indian market face challenges related to classification interpretation, documentation preparation, and CDSCO query responses. Businesses that require expert regulatory support can explore professional medical device import license services in India offered by Regacats Solutions to ensure accurate classification assessment and smoother approval.
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import License, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import License, and FSSAI regulatory consulting in India.
















