Page Contents
ToggleHow Foreign Manufacturers Can Obtain Medical Device Import License in India
India has become one of the fastest-growing medical device markets in the world. As a result, many global healthcare companies are expanding their presence in the country. However, before entering the Indian market, international companies must obtain a Medical Device Import License in India for Foreign Manufacturers in accordance with the Medical Device Rules, 2017 regulated by the Central Drugs Standard Control Organization (CDSCO).
This regulatory approval ensures that imported medical devices meet safety, quality, and compliance standards before reaching hospitals and healthcare providers in India.
Therefore, foreign manufacturers cannot legally import medical devices into India without CDSCO approval. The regulatory pathway involves appointing an Indian Authorized Agent, preparing regulatory documentation, submitting the MD-14 application, and obtaining the MD-15 import license.
Understanding this process is essential for international manufacturers planning to enter the Indian healthcare market. In this guide, we explain the complete process for obtaining a Medical Device Import License in India for Foreign Manufacturers, including documentation requirements, approval timelines, regulatory steps, and compliance obligations.
Additionally, experienced CDSCO regulatory consultants such as Regacats Solutions assist foreign manufacturers in navigating the CDSCO regulatory framework and obtaining MD-15 import license approvals efficiently.
Medical Device Import License in India for Foreign Manufacturers: Regulatory Overview
The Medical Device Import License in India for Foreign Manufacturers is issued by CDSCO to permit international companies to import and market medical devices in India.
India regulates medical devices under the Medical Device Rules, 2017, which establish regulatory pathways for manufacturing, importing, and distributing medical devices.
According to these regulations, foreign manufacturers must apply for an Medical Device import license through an Indian Authorized Agent.
The regulatory approval process involves two main application forms:
Form MD-14 – Application for import license
Form MD-15 – Import license granted by CDSCO
The MD-14 application is submitted online through the CDSCO portal along with supporting documents. After review and approval, CDSCO issues the MD-15 license.
This license allows foreign manufacturers to:
Import medical devices legally into India
Supply devices to distributors and hospitals
Participate in healthcare tenders
Expand business operations within the Indian healthcare market
Before submitting the application, manufacturers must ensure that their devices meet quality standards and regulatory documentation requirements defined by CDSCO.
Manufacturers must also determine the correct device classification before applying for an import license.
You can learn more about this in our guide on Medical Device Classification in India
What is the Need of Medical Device Import License in India for Foreign Manufacturers
Foreign manufacturers must obtain a Medical Device Import License India for Foreign Manufacturers before selling medical devices in India.
India’s healthcare regulatory framework prioritizes patient safety and product quality. Therefore, regulators require companies to demonstrate compliance with regulatory standards before introducing devices into the market.
There are several important reasons why this license is mandatory.
First, CDSCO evaluates the safety and effectiveness of medical devices before approving imports. This ensures that medical devices used in hospitals and healthcare facilities meet international quality standards.
Second, the import license allows regulatory authorities to monitor imported medical devices and maintain traceability within the supply chain.
Third, regulatory approval ensures compliance with Indian laws governing medical device import and distribution.
Companies that attempt to import medical devices without CDSCO approval may face legal penalties, product seizures, or market restrictions.
Therefore, obtaining the correct regulatory license becomes a critical step for foreign manufacturers planning to expand their business in India.
Many international companies work with experienced medical device import license consultants in India to ensure accurate documentation and smooth regulatory approval.
Step-by-Step Process for Medical Device Import License India for Foreign Manufacturers
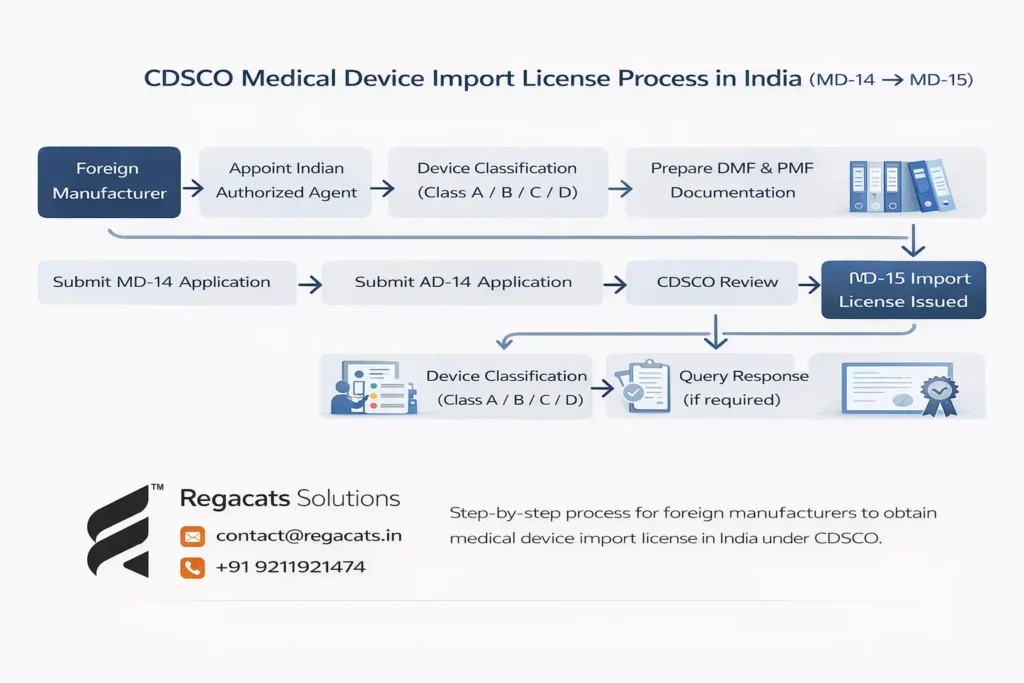
Foreign manufacturers must follow a structured regulatory pathway when applying for the Medical Device Import License in India for Foreign Manufacturers.
Understanding the CDSCO import license process helps manufacturers avoid regulatory delays and ensure successful approval.
Below is the standard regulatory process.
Step 1: Appointment of Indian Authorized Agent for Medical Device Import License in India for Foreign Manufacturer
Foreign manufacturers must appoint an Indian Authorized Agent to represent them before CDSCO.
The authorized agent acts as the official regulatory representative responsible for submitting applications and communicating with regulators.
Additionally, the agent must hold a valid drug license or wholesale license in India.
Without appointing an authorized agent, foreign manufacturers cannot submit an MD-14 application.
Step 2: Medical Device Classification

Before submitting the import license application, the medical device must be classified according to risk category.
Medical devices in India fall into four regulatory classes:
Class A – Low risk devices
Class B – Low to moderate risk devices
Class C – Moderate to high risk devices
Class D – High risk devices
The classification determines the regulatory review process and documentation requirements.
You can read our detailed guide on Medical Device Classification in India to understand how CDSCO categorizes medical devices.
Step 3: Preparation of Regulatory Documentation for Medical Device Import License in India for Foreign Manufacturer
After determining the device classification, manufacturers must prepare regulatory documentation required for submission.
These documents demonstrate device safety, manufacturing quality, and regulatory approvals.
Key documents include:
Device Master File (DMF)
Plant Master File (PMF)
Free Sale Certificate
ISO 13485 certification
Regulatory approvals from other countries
Product technical specifications
Labeling and packaging details
You can review the complete checklist of documents required for medical device import license in India.
Proper documentation significantly improves the chances of faster CDSCO approval.
Step 4: Submission of MD-14 Application for Medical Device Import License in India for Foreign Manufacturer
The Indian Authorized Agent submits the application through the CDSCO online portal using Form MD-14.
The application must include:
Regulatory documents
Device details
Manufacturing information
Government fee payment
Once submitted, CDSCO begins the technical review of the application.
Step 5: CDSCO Review and Queries
During the review process, CDSCO evaluates device safety data, regulatory approvals, and manufacturing standards.
Sometimes regulators request clarification or additional documents.
Responding to CDSCO queries quickly helps avoid delays in the approval process.
Experienced regulatory consultants often assist manufacturers with documentation and query responses.
Step 6: Grant of MD-15 Import License for Medical Device Import License in India for Foreign Manufacturer
After successful evaluation, CDSCO grants the MD-15 Import License.
This approval allows foreign manufacturers to legally import medical devices into India and distribute them through authorized channels.
Common Challenges in Medical Device Import License India for Foreign Manufacturers
Foreign manufacturers often face regulatory challenges when applying for the Medical Device Import License in India for Foreign Manufacturers.
For example, incorrect device classification can delay the application review process. Similarly, incomplete documentation frequently leads to CDSCO queries.
Additionally, manufacturers may struggle to understand regulatory procedures in India, which differ significantly from regulatory systems in the United States or European Union.
Therefore, many global manufacturers partner with regulatory consultants who specialize in CDSCO compliance and medical device regulations in India.
These experts guide companies through documentation preparation, regulatory submission, and query management.
Timeline and Cost for Medical Device Import License India for Foreign Manufacturers
The approval timeline for the Medical Device Import License in India for Foreign Manufacturers generally ranges between 6 to 9 months.
However, several factors influence the timeline, including:
Device classification
Documentation completeness
CDSCO review workload
Regulatory queries raised by authorities
Manufacturers should also understand the government fees associated with the MD-14 application.
You can read our complete guide on Medical Device Import License cost and timeline in India to understand regulatory fees and approval timelines.
Submitting well-prepared applications significantly reduces regulatory delays.
How Regacats Solutions Supports Foreign Manufacturers for CDSCO Medical Device Import License
Entering the Indian medical device market can be complex for foreign manufacturers. Regulatory requirements, documentation standards, and CDSCO procedures require specialized expertise.
Regacats Solutions provides comprehensive regulatory consulting services to help international manufacturers obtain CDSCO approvals efficiently.
The company supports global clients with:
Medical Device Import License (MD-14 and MD-15)
Indian Authorized Agent services
Medical device classification assistance
Regulatory documentation preparation
CDSCO submission and approval management
Regulatory query response and compliance support
By working with experienced regulatory consultants, manufacturers can reduce approval delays and ensure full compliance with Indian regulatory requirements.
If you are planning to import medical devices into India, Regacats Solutions provides end-to-end consulting services for obtaining Medical Device Import License (MD-14 and MD-15).
FAQs – MD Import License for Foreign Manufacturers
Conclusion
India offers tremendous growth opportunities for global medical device companies. However, manufacturers must comply with CDSCO regulatory requirements before entering the market.
Obtaining the Medical Device Import License India for Foreign Manufacturers remains a mandatory regulatory step for importing medical devices into India.
The approval process includes appointing an Indian Authorized Agent, preparing regulatory documentation, submitting the MD-14 application, and obtaining the MD-15 license.
Although the regulatory process may appear complex, proper preparation and expert regulatory guidance significantly simplify the pathway.
Foreign manufacturers that successfully obtain CDSCO approval can access one of the fastest-growing healthcare markets in the world.
With the support of experienced CDSCO consultant such as Regacats Solutions, international manufacturers can navigate the regulatory process smoothly and launch their medical devices in the Indian market with confidence.
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import License, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import License, and FSSAI regulatory consulting in India.
















