CDSCO Import License Consultant in India for Cosmetics, Medical Devices & Drugs
Table of Contents
ToggleCDSCO Import License Support for Cosmetics, Medical Devices & Drugs
India requires regulatory authorization before companies can import and distribute cosmetics, medical devices, or pharmaceutical products. The
Central Drugs Standard Control Organization (CDSCO) regulates these approvals under Indian law.
Therefore, whether you are a foreign manufacturer exporting to India or an Indian importer sourcing regulated products, you must obtain a CDSCO Import License before commercialization.
Regacats Solutions is a premium CDSCO Import License Consultant based in Gurugram, Haryana, with:
8+ years of focused regulatory experience
1000+ applications supported
Structured compliance methodology
Strong record of timely approvals
We provide end-to-end regulatory consulting for:
Cosmetics Import Registration (COS-2)
Medical Device Import License (MD-14 / MD-15)
Drug Import License
Because Indian regulatory compliance demands documentation precision and structured filing, we align your application with CDSCO expectations from the beginning.
What Is a CDSCO Import License?
A CDSCO Import License is mandatory regulatory authorization required to legally import:
Cosmetic products
Notified medical devices
Pharmaceutical products and drugs
CDSCO grants approval under:
Drugs & Cosmetics Act, 1940
Medical Device Rules, 2017
Cosmetic Rules, 2020
Without proper import authorization:
Customs authorities may detain shipments
Distribution rights may be restricted
Market entry may be delayed
Financial risks may increase
As a result, structured regulatory planning protects your commercial timeline and brand reputation.
Why You Need a CDSCO Import License Consultant
Although companies can technically initiate the application process, CDSCO approvals involve detailed documentation, classification accuracy, and regulatory interpretation.
A professional CDSCO Import License Consultant helps you:
Identify correct product classification
Align international documents with Indian requirements
Structure the application strategically
Avoid repeated deficiency cycles
Reduce approval delays
Because regulatory gaps often arise from format misalignment rather than product quality, expert guidance significantly improves approval efficiency.
About Regacats Solutions – Premium CDSCO Import License Consultant

Regacats Solutions operates from Gurugram, Haryana and specializes exclusively in CDSCO import approvals.
Unlike general regulatory firms, we focus on:
Cosmetics Import License India
Medical Device Import License India
Drug Import License India
Over the past 8 years, we have supported more than 1000 applications across diverse product categories. Consequently, we follow a defined Import Compliance Framework that ensures transparency, predictability, and disciplined execution.
We assist:
Manufacturers from any country exporting to India
Indian importers and distributors
Global brand owners entering the Indian market
Regulatory firms seeking Indian representation
Furthermore, we align documentation prepared under US FDA, EU CE, ISO 13485, and GMP standards with Indian regulatory formats.
Our CDSCO Import Services
Cosmetics Import License (COS-2 Registration)
Medical Device Import License (MD-14 / MD-15)
Drug Import License
INDIAN AUTHORIZED AGENT (IAA) SERVICES
Foreign manufacturers must appoint an Indian Authorized Agent to obtain CDSCO licenses.
Regacats Solutions provides independent and compliant IAA representation for:
Cosmetics
Medical Devices
Drugs & Pharmaceutical Products
Cosmetics Import License (COS-2 Registration)
This applies to:
Skincare products
Haircare formulations
Makeup and color cosmetics
Personal care products
Our Cosmetics Approval Process
First, we verify ingredient compliance under Indian standards.
Next, we review labeling conformity.
Then, we assess manufacturer documentation and Free Sale Certificates.
After that, we prepare and submit the application through the SUGAM portal.
Finally, we manage CDSCO queries until approval.
Because cosmetic reviews focus heavily on ingredient permissibility and labeling accuracy, early evaluation reduces regulatory objections.
Medical Device Import License (MD-14 / MD-15)
Devices fall under Class A, B, C, or D depending on risk category.
Our Device Licensing Framework
We begin with classification confirmation.
Subsequently, we conduct a technical file gap analysis.
In addition, we review CE marking and ISO 13485 certifications.
Thereafter, we prepare and file MD-14 applications.
Finally, we coordinate until MD-15 license issuance.
Because classification accuracy determines regulatory scrutiny level, early assessment improves approval timelines.
Drug Import License
This includes:
Finished formulations
APIs
OTC medicines
Specialty pharmaceuticals
Our Pharmaceutical Import Strategy
Initially, we determine the regulatory pathway.
Then, we review GMP documentation and Site Master File details.
Next, we assist with Form 8 and Form 10 preparation.
After submission, we manage technical queries.
Ultimately, we coordinate until import approval is granted.
Because pharmaceutical applications undergo detailed evaluation, structured dossier preparation enhances approval predictability.
Our Structured CDSCO Import License Process
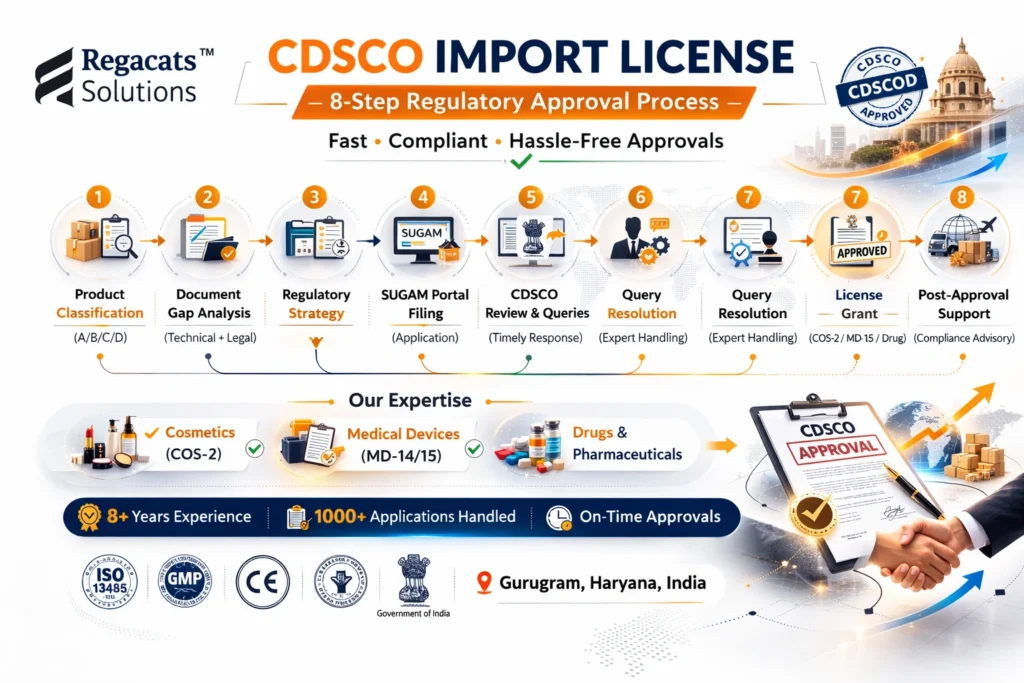
To maintain clarity and accountability, we follow a defined 8-step framework:
Product Classification
Documentation Gap Analysis
Regulatory Strategy Development
Dossier Compilation
SUGAM Portal Filing
CDSCO Query Resolution
Import License Grant
Post-Approval Compliance Advisory
Because consistency drives regulatory efficiency, our structured approach minimizes delays and ensures measurable progress.
Frequently Asked Questions
What is CDSCO Import License?
A CDSCO Import License is mandatory regulatory approval required to legally import cosmetics, medical devices, or pharmaceutical products into India.
Who can apply for CDSCO Import License?
Foreign manufacturers and Indian importers can apply through an authorized Indian entity.
How long does CDSCO Import License approval take?
Approval generally takes 3–6 months depending on product category and documentation quality.
Why hire a CDSCO Import License Consultant?
A consultant reduces documentation errors, improves compliance alignment, and accelerates regulatory approval.
Start Your CDSCO Import License Process with Regacats Solutions
India’s regulatory environment requires disciplined documentation and proactive compliance planning.
If you are planning to import or export regulated products into India, Regacats Solutions provides premium CDSCO Import License consulting designed for timely and structured approvals.
Gurugram, Haryana, India
Email: contact@regacats.in
Phone: +91 9211921474
Schedule a CDSCO Import License Consultation Today
Receive a structured compliance roadmap tailored to your product category.
















