FSSAI Consultant for Nutraceutical Import in India
Table of Contents
ToggleFSSAI Nutraceutical Import Consultant for Global Brands & Indian Importers
As a leading FSSAI Nutraceutical Import Consultant, Regacats Solutions provides complete regulatory and compliance support for nutraceutical and dietary supplement imports into India. We assist foreign manufacturers, global supplement brands, and Indian importers in securing FSSAI Central licenses, validating ingredient permissibility, ensuring labeling compliance, and managing port-level clearances.
Because India enforces strict food safety regulations, importing nutraceuticals without proper compliance planning can result in shipment detention, rejection, or financial loss. Therefore, businesses rely on an experienced FSSAI Consultant for compliance of Dietary Supplement & Nutraceutical to navigate approvals efficiently and minimize regulatory risk.
Why You Need an FSSAI Nutraceutical Import Consultant in India
India’s nutraceutical sector operates under multiple regulatory frameworks. Consequently, importers must comply with:
Food Safety and Standards Act, 2006
Labelling & Display Regulations
Advertising & Claims Regulations
Non-Specified Food (NSF) Regulations
An experienced FSSAI Consultant help importer, global brands & foreign manufacturer to enter Indian market with ease for Nutraceutical & Dietary Supplement products ensures that your product meets all regulatory requirements before shipment dispatch.
Moreover, early regulatory planning significantly reduces clearance delays at Indian ports.
What an FSSAI Nutraceutical Import Consultant Actually Does
An experienced FSSAI Consultant for Nutraceutical & Dietary Supplement products can help Importers, global brands & foreign manufacturer to enter Indian market by :
- Regulatory classification assessment
- Ingredient and dosage validation
- FSSAI Central License application
- Import endorsement support
- Label compliance adaptation for India
- Non-Specified Food (NSF) approval (if required)
- Pre-shipment compliance audit
- Port clearance coordination
In short, an experienced FSSAI Consultant of Nutraceutical & Dietary Supplement products converts regulatory complexity into a predictable Indian Market entry pathway.
Step-by-Step Process Managed by an FSSAI Consultant in Nutraceutical Import
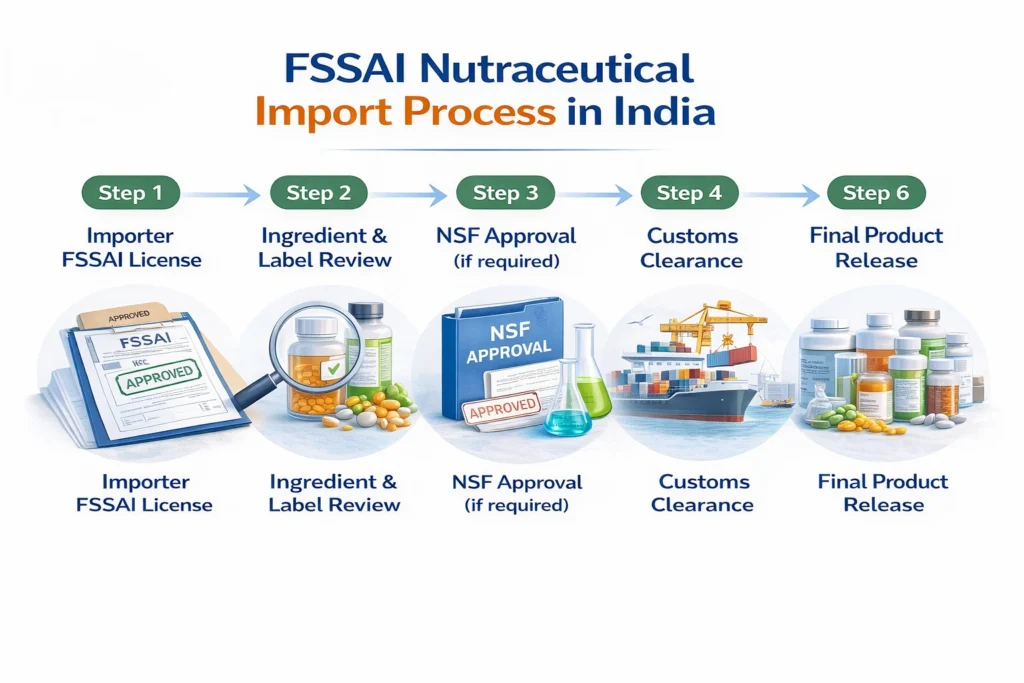
Step 1: Regulatory Classification of product by FSSAI Consultant before import of Nutraceutical
First, we determine whether your product qualifies as:
Nutraceutical
Dietary supplement/Health Supplement
Food for Special Dietary Use
Food for Special Medical Purpose
Prebiotic or Probiotic Food
Accurate classification prevents downstream objections.
Step 2: Ingredient & Dosage Validation by FSSAI Consultant before import of Nutraceutical
Next, our FSSAI Nutraceutical & Dietary Supplement product Consultant team verifies:
Ingredient inclusion in FSSAI schedules
Permissible vitamin and mineral limits
Botanical standardization compliance
Additive usage alignment
Prohibited substance screening
If ingredients fall outside permitted lists, we initiate NSF approval.
Step 3: FSSAI Central License for Importer
Importers must hold a valid FSSAI Central License with import endorsement. Therefore, our FSSAI Nutraceutical & Dietary Supplement product Consultant prepares documentation, files the application, and coordinates authority responses.
Step 4: Label Adaptation for Indian Market
Imported nutraceutical labels must include:
FSSAI logo
FSSAI license number
Veg/Non-Veg symbol
Nutritional table format as per Indian norms
Allergen declaration
RDA compliance statements
Importer name and address
Country of origin
Mandatory disclaimers
Before shipment dispatch, our FSSAI Nutraceutical & Dietary Supplement Consultant team reviews artwork to eliminate compliance gaps.
Step 5: Non-Specified Food (NSF) Approval (If Required)
If your ingredient is not listed in FSSAI schedules, prior approval becomes mandatory.
Our team prepares:
Scientific safety justification
Toxicology evidence (if applicable)
Exposure assessment
Global regulatory references
Structured dossier submission
Because NSF approvals require scientific reasoning, professional support accelerates review cycles.
Step 6: Foreign Manufacturing Facility Registration (If Applicable)
Certain high-risk categories require foreign facility registration before export. We coordinate this process seamlessly.
Step 7: Pre-Shipment Compliance Audit
Before shipping products to India, we conduct:
Documentation verification
COA alignment review
Label compliance confirmation
Claim validation check
This proactive review prevents costly port rejections.
Step 8: Port-Level Import Clearance
Below is a visual representation of the FSSAI nutraceutical import clearance process managed by Regacats Solutions.

Upon arrival:
FSSAI may draw samples
Laboratory testing may be conducted
Clearance is issued upon compliance
Our FSSAI Nutraceutical Import Consultant coordinates communication with customs brokers and FSSAI officers to ensure smooth clearance.
Common Challenges in Nutraceutical Import to India
Without professional guidance, importers often face:
- Excess vitamin dosage rejection
- Ingredient non-permissibility
- Label misalignment
- Claim overstatement
- Missing documentation
- Incorrect HS classification
- Shipment detention
However, with a structured FSSAI Nutraceutical Import Consultant strategy, these risks reduce significantly.
Documents Required for Nutraceutical Import into India
Prepare the following early:
FSSAI Central License copy
IEC (Import Export Code)
Product composition
Certificate of Free Sale
Raw material COA
Country of origin certificate
Manufacturing license copy
Label artwork
Power of Attorney (if applicable)
Our FSSAI Nutraceutical Import Consultant organizes documentation in authority-ready format.
Estimated Timelines
- Central License: 2–4 weeks
- Label compliance review: 3–5 days
- NSF Approval: 4–6 months
- Port clearance: 2–12 days
Timelines vary based on product complexity and scientific evaluation requirements.
Why Global Brands Choose Regacats as Their FSSAI Nutraceutical Import Consultant
- 8+ years of regulatory specialization
- Dedicated nutraceutical compliance expertise
- Experience with global dietary supplement brands
- Structured dossier preparation
- Transparent documentation approach
- Strong authority coordination
We operate as your regulatory bridge between international manufacturing facilities and Indian food authorities.
Frequently Asked Questions
Is FSSAI approval required before importing nutraceuticals?
Yes. Importers must ensure licensing, label compliance, and ingredient validation before shipment.
Do foreign brands need Indian representation?
Often yes. Depending on structure, regulatory coordination through an Indian entity becomes necessary.
What happens if shipment fails testing?
FSSAI may order re-export, destruction, or corrective action. Therefore, pre-shipment compliance is critical.
Partner with Regacats Solutions for import of Nutraceutical Products
Importing nutraceuticals into India demands scientific validation, documentation accuracy, and regulatory alignment. With Regacats Solutions as your FSSAI Nutraceutical Import Consultant, you reduce delays, avoid costly rejections, and accelerate compliant market entry.


















