Page Contents
ToggleMedical Device Import License Cost & Timeline in India (MD15 Guide)
Understanding the medical device import license cost in India is essential before entering the Indian healthcare market. Every importer and foreign manufacturer must evaluate the total charges of medical device import license, government fees, and approval timeline before submitting an MD14 application. Therefore, proper budgeting and regulatory planning directly influence commercial success.
In this guide, we explain:
Medical device import license structure
Government fees under MDR 2017
Professional consultancy charges
MD15 approval timeline
Factors that increase cost or delay approval
This article applies specifically to medical devices regulated under CDSCO.
What Is the Medical Device Import License Cost in India?
The total charges of medical device import license depends on multiple regulatory and operational factors. While CDSCO prescribes fixed government fees, the overall MD import license cost varies based on class, number of devices, and manufacturing sites.
Generally, the cost includes:
Government fees
Documentation preparation expenses
Authorized Agent charges
Professional consultancy fees
Moreover, higher-risk devices often require more technical documentation, which increases preparation effort and overall cost.
Government Fees for Medical Device Import License
CDSCO charges government fees under the Medical Device Rules, 2017. These fees apply:
Per Device Class
Per manufacturing site
Per distinct device
Class C and Class D devices require deeper technical review. Consequently, documentation preparation becomes more extensive, which indirectly increases the effective import cost of medical device.
Before submission, applicants must calculate fees accurately. Otherwise, incorrect fee payment may trigger avoidable delays.
Professional Charges and Consultancy Cost
Although government fees remain fixed, professional CDSCO Consultant charges vary depending on regulatory complexity.
Consultancy fees usually depend on:
Device classification
Number of SKUs
Documentation completeness
Query handling requirements
Authorized Agent services
Experienced consultants reduce documentation errors and respond to regulatory queries efficiently. As a result, applicants avoid repeated submissions and unnecessary delays.
If you require structured regulatory support, you may review our medical device import license consultant in India services for end-to-end assistance.
Medical Device Import License Timeline in India
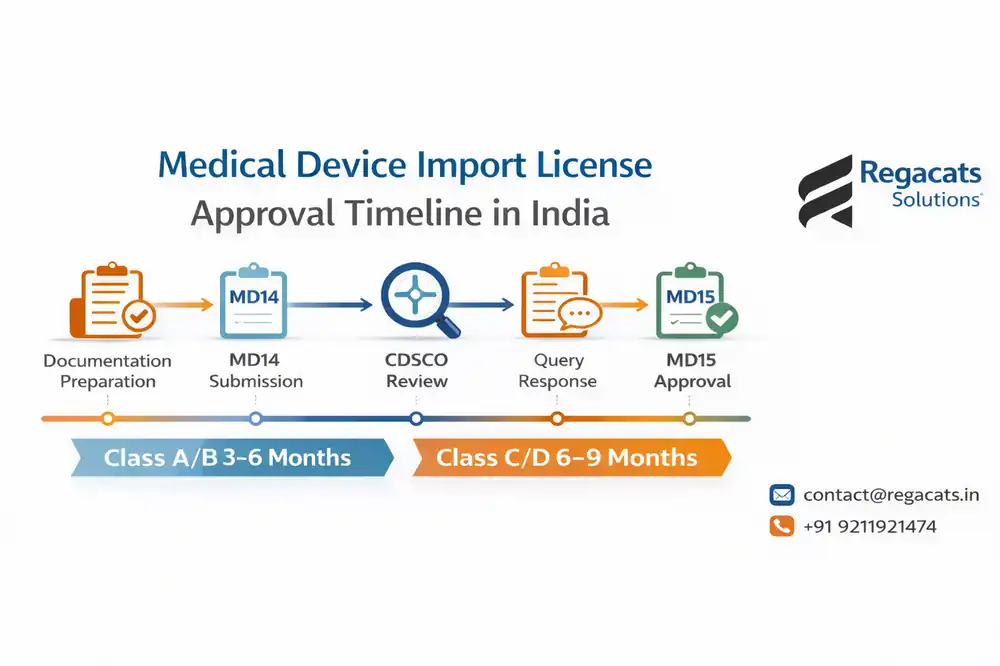
While understanding import cost of medical device license is important, timeline planning is equally critical.
Class A & B Devices
Approval typically takes 3 to 6 months, provided documentation meets CDSCO standards.
Class C & D Devices
Approval generally takes 6 to 9 months, as CDSCO conducts detailed technical evaluation.
However, applicants who submit incomplete Device Master Files often face extended review periods. Therefore, technical accuracy directly influences approval speed.
You may review Medical Device Import License India (MD-15) | CDSCO Guide for complete guide on import of Medical Device in India
Factors That Increase Medical Device Import License Cost
Several issues may increase overall importing cost of medical device license or delay approval:
Incorrect device classification
Incomplete Device Master File
Improper labeling compliance
Weak Free Sale Certificate
Delayed response to CDSCO queries
Furthermore, regulatory resubmissions increase both time and professional costs. Consequently, proactive documentation planning reduces financial and operational risks.
How to Reduce Medical Device Import License Cost and Delays
Importers and foreign manufacturers can reduce medical device import license cost and approval risks by:
Conducting classification review before filing
Preparing structured technical documentation
Ensuring MDR 2017 labeling compliance
Appointing an experienced Indian Authorized Agent
Responding to CDSCO queries promptly
Therefore, strategic regulatory preparation improves approval probability and reduces avoidable expenditure.
Many companies entering India partner with regulatory specialists who manage documentation and compliance proactively. If you plan to import medical devices into India, you may explore our authorized agent services for medical devices in India for structured regulatory assistance.
Frequently Asked Questions – Medical Device Import License Cost & Timelines
Conclusion
The import cost of Medical device license and approval timeline directly impact market entry strategy in India. While government fees remain structured under MDR 2017, documentation quality and regulatory handling significantly influence total cost and review duration.
Therefore, importers and foreign manufacturers should approach the process with technical accuracy and compliance planning.
If you require assistance with classification, documentation preparation, MD14 filing, or authorized agent appointment, explore our medical device import license services in India for comprehensive regulatory support.
About Regacats Solutions
Regacats Solutions is an India-based regulatory consulting firm specializing in CDSCO cosmetic import licensing, medical device import registration, and FSSAI regulatory compliance. With extensive experience supporting Indian importers and global cosmetic brands, we assist with COS-1 and COS-2 registration, labeling compliance, Legal Metrology requirements, and post-approval regulatory obligations. Our team focuses on India-specific compliance frameworks to ensure faster approvals, accurate documentation, and long-term regulatory compliance.
Content Reviewed by
Regulatory Experts at Regacats Solutions
Specialists in CDSCO cosmetic import licensing, Legal Metrology compliance, EPR authorization, medical device import registration, and FSSAI regulatory consulting in India.
Need Help with CDSCO Import License in India?
If you are an importer or foreign manufacturer planning to register medical devices in India, professional regulatory assistance can simplify the entire MD15 process.
Explore our detailed service offering here:
CDSCO Medical Device Import License (MD-14, MD-15) Services in India
Talk to a Regulatory Expert Now
Contact:
Email: contact@regacats.in — Phone: +91 9211921474 — Get a free consultation
















